Meet the
SonoStik™
team
Together, we bring decades of clinical and commercial expertise to our mission, product, and business.

Together, we bring decades of clinical and commercial expertise to our mission, product, and business.

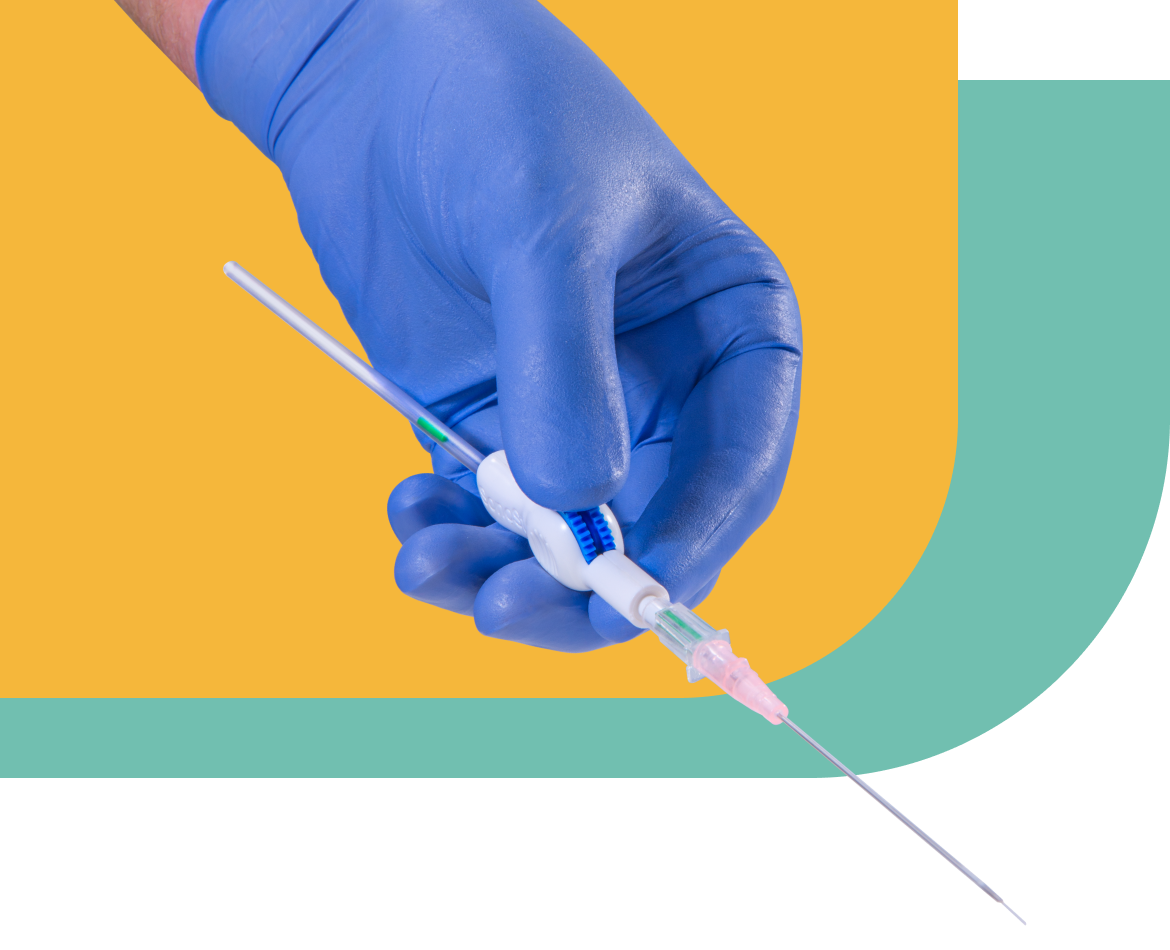

In 2013, a team of physicians at George Washington Hospital found a new purpose: solving a persistent clinical challenge they faced nearly every day.
Their innovation: a device that turns even the most difficult peripheral stick into a quick, one-handed procedure, enabling their fellow clinicians to achieve first-try success for more patients than ever.
Our product experts have extensive MedTech expertise.
Our commercial leaders know what it takes to achieve success.
Our innovators have launched and scaled multiple new ventures.

Co-Founder, Chairperson of SonoStik BoD
One of the most respected experts in her region, Dr. Almansouri is currently Consultant Endocrinologist and Deputy Medical Director of the Imperial College London Diabetes Centre in the UAE.
In addition to her clinical role, Her Excellency regularly appears at high-profile forums in the UAE and internationally as a speaker, chairperson, and emcee. She is heavily involved in UAE-based initiatives to promote STEM education, empower Emirati women, and encourage enterprise by Emirati youth.
Dr. Almansouri is also the first Emirati to be awarded the Presidential Scholarship to study medicine in the US, a 2014 recipient of the “UAE Pioneers Award,” as well as the first non-American woman to graduate from George Washington University School of Medicine and Health Sciences.

Co-Founder, Chief Medical Officer
Both an experienced clinician and noted innovator, Dr. Sikka is a board-certified emergency medicine physician, Professor of Emergency Medicine at the George Washington University School of Medicine and Health Sciences, and Chief of the Innovative Practice & Telemedicine Section for The GW Medical Faculty Associates.
He is also an active researcher focused on improving patient care through technology, and has received funding from the McKesson Foundation and the Centers for Medicare and Medicaid Health Care Innovation Challenge Grant Award. A committed clinical educator, Dr. Sikka has developed both undergraduate and graduate curricula related to emergency medicine and telemedicine, and regularly speaks at regional and national meetings on related topics.

Co-Founder, Director of Medical R&D
One of the original developers of the SonoStik concept, Dr. Corman is a board-certified emergency medicine physician who specializes in the development and application of ultrasound technology in the acute care setting.
He completed his medical residency training at The George Washington University Medical Center, and is now a nationally recognized clinician and ultrasound expert. He has honed his skills as both a clinician and innovator at nearly a dozen large regional medical centers throughout the US.

Head of Scientific Advisory Committee
A leading expert in his field, Dr. Schears is a board-certified Assistant Professor of Anesthesiology at the Mayo Clinic, as well as a consultant to the Mayo Clinic’s Department of Anesthesiology. As an advisor to SonoStik, he brings a wealth of clinical and commercial expertise to our product development and marketing efforts.
In addition to his clinical pursuits, Dr. Schears is the founder of Lumen-Vue, a groundbreaking startup developing technology that uses NIR to determine catheter tip location. He also serves as a scientific advisor for numerous other vascular access and visualization companies.

Chief Executive Officer
A veteran executive with deep roots in vascular access, Gary has more than two decades of experience in building and managing companies like SonoStik. His expertise includes raising capital, building advisory boards with nationally known key opinion leaders, and successfully bringing disruptive technologies to market.
Throughout his career, he has served as a leader, advisor, and consultant for numerous MedTech startups, and also managed successful territories, regions, and divisions for devices in the critical care, cardiac, and central venous access markets. His current focus: helping innovators like SonoStik perfect their product/market fit, mature their operations, and scale their sales.

Commercialization & Operations

Chief Technology Officer, BoD Member
An accomplished innovator and original member of the SonoStik team, Benjamin is the engineering pioneer who helped design and fully realize our first device prototype. In addition to his work with SonoStik, he is also the founder of two other successful medical device companies and the author of 7 published patents related to medical device designs (excluding additional filings).
Dr. Holmes holds a Ph.D. in Mechanical and Aerospace Engineering from George Washington University, with a focus on novel biomedical implants and devices.

Operations Director
The operational expert behind our development and launch activities, Peter brings four decades of strategic financial strategy and business development experience to our team – as well as more than 25 years of experience developing long-range business plans at all corporate levels.
He has extensive experience developing tender-based business for an enterprise with up to $500M in annual revenue, and has effectively and successfully managed annual budgets of up to $12M.
With the launch of SonoStik, our team will introduce an innovation with the potential to enhance core clinical protocols in nearly every department that relies on efficient, timely IV placement.
The market for peripheral venous access products is growing rapidly, with nearly $7BN in annual sales and a 6% CAGR. SonoStik will be a truly differentiated new addition — one that represents both an important clinical advance and a significant financial opportunity.
SonoStik has been successfully reviewed and cleared by the FDA.
Reference: 1. Vital Signs: Central line-associated blood stream infections—US.,2001,2008.2009. Morb. Mortal Wkly. Rep. 2011: 60:243-8

Globally recognized clinical specialist and trailblazer
One of the most respected experts in her region, Dr. Almansouri is currently Consultant Endocrinologist and Deputy Medical Director of the Imperial College London Diabetes Centre in the UAE.
In addition to her clinical role, Her Excellency regularly appears at high-profile forums in the UAE and internationally as a speaker, chairperson, and emcee. She is heavily involved in UAE-based initiatives to promote STEM education, empower Emirati women, and encourage enterprise by Emirati youth.
Dr. Almansouri is also the first Emirati to be awarded the Presidential Scholarship to study medicine in the US, a 2014 recipient of the “UAE Pioneers Award,” as well as the first non-American woman to graduate from George Washington University School of Medicine and Health Sciences.